Tanso Biosciences has developed a high-performance functional assay to overcome the barriers in GPCR drug discovery. This cutting-edge technology enables functional assays of all GPCRs regardless of coupled G-protein subtypes, making it a powerful tool in drug discovery enterprises.
- 1. About GPCRs
- 2. Core Technology
- 2.1. Key Features
- 2.2. Components
- 2.2.1. Proprietary Assay Cells
- 2.2.2. Gα protein cocktail
- 2.2.3. Comprehensive GPCR library
- 3. Validation
- 3.1. AGTR1, a peptide-ligand receptor
- 3.2. GPR91, a low molecular ligand receptor
- 3.3. Flt3, a receptor tyrosine kinase (RTK)
- 3.4. Reproducibility
- 4. Conclusion
- 5. FAQs
About GPCRs
G-protein-coupled receptors (GPCRs) are a highly diverse class of membrane receptors that play critical roles in human physiology. With approximately 800 members in the human genome, they are the largest group of receptors and regulate a wide range of biological processes, including blood pressure, blood glucose levels, and cognitive functions. Out of these 800 GPCRs, approximately 200 respond to endogenous mediators, while the remaining 500 function as sensory receptors, detecting smells, tastes, and other stimuli. The last 100 GPCRs remain unclassified, with their ligands yet to be identified.
GPCRs have been a prime target for drug discovery, particularly receptors for endogenous mediators. One-third of drugs in the market target GPCRs. It is because many diseases, such as cardiovascular and neurological disorders, involve the dysregulation of these receptors. In addition, recent studies have shown that orphan and sensory receptors also play important roles in various pathophysiology. These findings highlight the vast untapped potential of the majority of GPCRs in the field of drug development.
Despite their promise, however, only a small portion of GPCRs have drugs developed to target them. The lack of adequate and reliable assays has been a barrier to GPCR drug discovery. Current binding assays suffer from false positives and false negatives, and functional assays have limitations, such as being time-consuming, expensive, or limited in their applicability. Additionally, the G12/13 and Gi/o subtypes of G-proteins are notoriously intractable and further complicate GPCR research.
Tanso Biosciences has developed a high-performance functional assay to overcome these barriers and make high-hanging GPCRs accessible to the pharmaceutical industry. Our core technology combines sensitive and versatile reporter cells, a comprehensive Gα protein cocktail, and a comprehensive human and mouse GPCR library to make possible functional assays of all GPCRs.
Core Technology
Key Features
Our cell-based GPCR functional assay has the following key features:
- Remarkable sensitivity and signal-to-background (S/B) ratio
- An exhaustive library of 300 human GPCRs and 250 mouse orthologs, including both non-orphan and orphan receptors
- Works for all receptors coupled with any major Gα proteins, including Gq, Gs, Gi/o, and G12/13
- Variety of assay modes, including agonist, antagonist, positive allosteric modulator (PAM), and negative allosteric modulator (NAM)
Components
The core technology for our cell-based GPCR functional assay consists of sensitive and versatile test cells, a Gα protein cocktail, and an exhaustive human GPCR library.
Proprietary Assay Cells
Our proprietary assay cells are equipped with an artificial gene circuit that amplifies GPCR-induced intracellular signals, resulting in highly sensitive and specific results. These cells can work with all major Gα proteins, including Gq, Gs, Gi/o, and G12/13. The cells have been verified with 99% (208 out of 210) of non-orphan GPCRs. Furthermore, they have proved themselves in assays with other membrane receptors, such as receptor tyrosine kinases. The technology has been filed for international patent by The University of Tokyo and is exclusively licensed to Tanso Biosciences (Publication Number WO/2020/026979).
Gα protein cocktail
The all-around Gα protein cocktail complements our proprietary assay cells and enables functional assays for any GPCR under a single protocol, regardless of the Gα protein the target receptor is coupled with.
Comprehensive GPCR library
Our comprehensive GPCR library is an industry-leading collection of non-sensory GPCRs, using native sequences to minimize signal artifacts. The library includes 216 non-orphan human GPCRs and 94 orphan GPCRs, 187 non-orphan mouse GPCRs, and 63 orphan mouse GPCRs, totaling 300 human and 250 mouse receptors. Visit our GPCR Catalog for further details.
Validation
Our assay has yielded consistent readouts with results from existing methods and at high sensitivity and S/B ratio. Also, the results are consistent, whether tests are performed by different experimenters or on different days. In the figure, we measured EC50 values for 116 human GPCRs and compared them with EC50 or dissociation constants curated by IUPHAR.
We compared our assay and existing methods using a few representative GPCRs below.
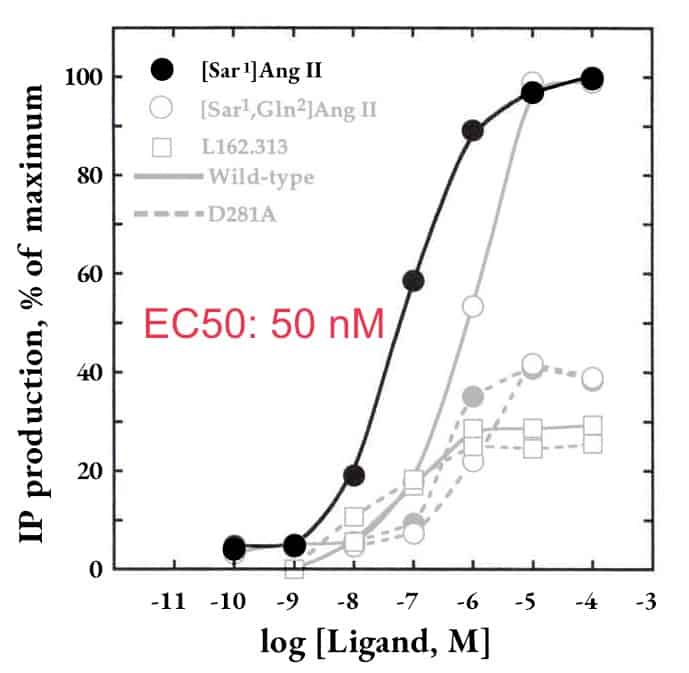
AGTR1, a peptide-ligand receptor
Tanso's method
Binding assay
| Ligand | Ki (nM) ± s.e.m. | n |
|---|---|---|
| Sar1-angiotensin ⅠⅠ | 0.38 ± 0.07 | 3 |
| Sar1, Ala8-angiotensin ⅠⅠ | 0.43 ± 0.08 | 3 |
| Sar1, Ile8-angiotensin ⅠⅠ | 0.46 ± 0.03 | 3 |
| Ile7-angiotensin ⅠⅠⅠ | 1.1 ± 0.3 | 3 |
| Angiotensin ⅠⅠ | 1.6 ± 0.2 | 3 |
| Angiotensin ⅠⅠⅠ | 5.4 ± 0.5 | 3 |
| Dup753 | 6.3 ± 0.9 | 3 |
| Angiotensin Ⅰ | 74 ± 12 | 3 |
| Arg8-vasopressin | >1,000 | 2 |
| Endothelin | >1,000 | 2 |
| PD123177 | >10,000 | 2 |
GPR91, a low molecular ligand receptor
In our assay, GPR91 responded to its agonist at high sensitivity and S/B ratio and consistently with the results from existing methods.
Tanso's method
Ca2+ assay
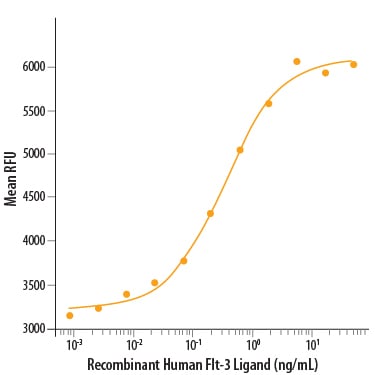
Flt3, a receptor tyrosine kinase (RTK)
Tanso's method

Cells from the same line of test cells were used in this assay as in assays for GPCRs, and Flt3 was transiently transfected into cells in the same way as GPCRs.
Reproducibility
Reproducibility is key when it comes to pharmaceutical research. Tanso Biosciences' GPCR functional assay is not only sensitive and versatile but highly reproducible. We conducted a series of experiments to verify the consistency of our assay, and the results were impressive.
First, we performed concentration-response analyses for β1 adrenoceptor (ADRB1) vs. isoproterenol on three different days (Figure 1, Table 1). Each test yielded a very close EC50 value, indicating excellent reproducibility of the assay.
This figure displays the results of three independent tests conducted on different days, in which ADRB1-expressing reporter cells were treated with the receptor agonist isoproterenol (n = 4, mean ± SEM). The data were fitted to sigmoid curves with a common Hill coefficient.
| Test | EC50 | Hill coefficient |
| 1 | 33 nM | 2.8 |
| 2 | 36 nM | 2.8 |
| 3 | 31 nM | 2.8 |
| Aggregate | 33 nM | 2.8 |
Next, we examined the variability between experimenters. Two experimenters in our lab conducted concentration-response analyses for type 1 angiotensin 2 receptor (AGTR1) and yielded comparable results both in the agonist mode (Figure 2, Table 2) and the antagonist mode (Figure 3, Table 3).
This figure displays the results of identical tests conducted by two experimenters, in which AGTR1-expressing reporter cells were treated with the receptor agonist angiotensin II (n = 4, mean ± SEM). The data were fitted to sigmoid curves with a common Hill coefficient.
| Experimenter | EC50 | Hill coefficient |
| 1 | 160 pM | 2.0 |
| 2 | 120 pM | 2.0 |
| Aggregate | 140 pM | 2.0 |
This figure displays the results of identical tests conducted by two experimenters, in which AGTR1-expressing reporter cells were treated with the receptor antagonist valsartan in the presence of angiotensin II (n = 4, mean ± SEM). The data were fitted to sigmoid curves with a common Hill coefficient.
| Experimenter | IC50 | Hill coefficient |
| 1 | 2.1 nM | -1.4 |
| 2 | 2.4 nM | -1.4 |
| Aggregate | 2.2 nM | -1.4 |
Overall, our verification process demonstrated that Tanso Biosciences' GPCR functional assay consistently produces reliable results, regardless of who conducts the test or when it is conducted. Trust in the reproducibility of our assay and choose Tanso Biosciences for your research needs.
Conclusion
In conclusion, Tanso Biosciences has made significant advancements in the field of GPCR research with its high-performance functional assay. This assay offers a solution to the current limitations in GPCR drug discovery, allowing for the development of drugs targeting all GPCRs, including both non-orphan and orphan receptors, coupled with any major Gα proteins. With its remarkable sensitivity and vast receptor coverage, this assay is poised to be a valuable tool in the pharmaceutical industry's pursuit of innovative drug development.
FAQs
What are GPCRs, and why are they important in drug discovery?
GPCRs are a class of membrane receptors that play a critical role in regulating various biological processes in the human body. With around 800 members in the human genome, they are the largest group of receptors. They are a prime target for drug discovery due to their role in regulating blood pressure, blood glucose levels, and many other functions.
What is Tanso Biosciences' cell-based GPCR functional assay?
Tanso Biosciences' cell-based GPCR functional assay is a high-performance assay that enables functional assays of all major GPCR types. It combines proprietary assay cells, a comprehensive Gα protein cocktail, and a comprehensive human GPCR library to make functional assays possible for all GPCRs, including non-orphan and orphan receptors, regardless of the subtype of coupled Gα proteins.
How does Tanso Biosciences' GPCR assay differ from other assays in the market?
Tanso Biosciences' functional assay has several key features that set it apart from other assays in the market. These include remarkable sensitivity and signal-to-background ratio, an exhaustive library of 550 human and mouse GPCRs, and the ability to work with all major Gα proteins, including Gq, Gs, Gi/o, and G12/13.
Do your reporter cells amplify noise as well as signals?
The artificial gene circuit in our cells is designed to amplify signals quadratically while boosting noise only by ~two-fold. The cells have responded to stimuli with >100-fold signal/background ratios and small standard errors in many GPCRs(see Validation).
Are your test cells capable of expressing orphan GPCRs in the plasma membrane when they are transfected?
It is essential to make sure that introduced orphan GPCRs are present in the plasma membrane all the more because they do not have agonists to prove that pharmacologically. We are testing each orphan receptor now.
Do you have animal GPCR panels?
Yes, we have mouse panels and are expanding our library to GPCRs in other species. If you are interested in a particular receptor, we can prioritize cloning it and will be ready to assay it in a few weeks.
Contact Us
Send us a message via the form below or write to us at [email protected].
Ordering Process
Service flow chart
Request a quotation
We hear about the study outline and propose experiments. A non-disclosure agreement is optional.
Place an order
You place an order, ship your compounds to our lab, and pay the fee via bank transfer or by credit card.
Wait
The turnaround time can be as short as two weeks, depending on the study scale and design and the backlog of orders.
Receive the report
You receive the study report in PDF.